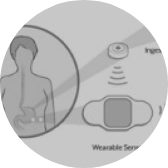
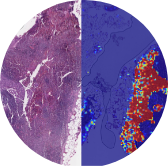
Major advances in digital are driving the MedTech and IoMT (Internet of Medical Things) industry.
A growing number of internet-connected medical devices create the ability to generate, collect, analyze, and transmit critical health data to improve care while gaining substantial operational efficiencies. The problem that companies face, however, is that to take advantage of this collected data, you need to have secure, high-performance device connectivity in the cloud that can provide actionable insights and, importantly, a patient-centric approach.